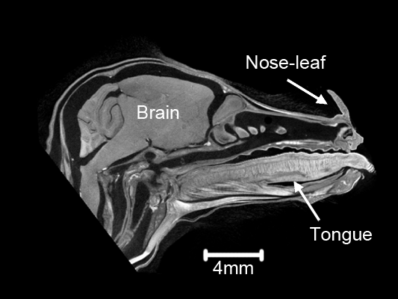
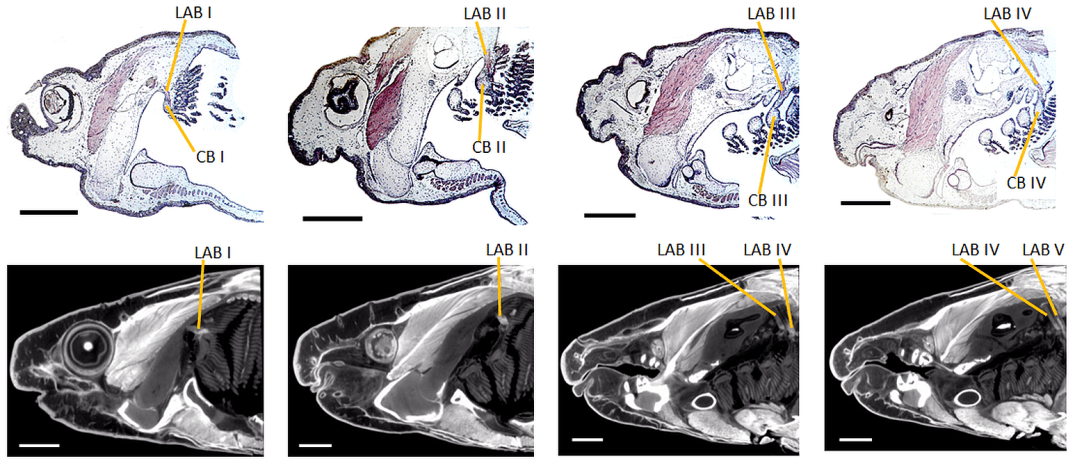
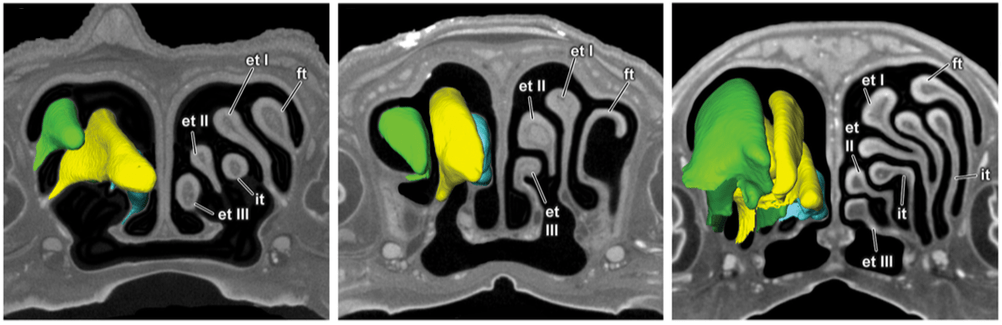
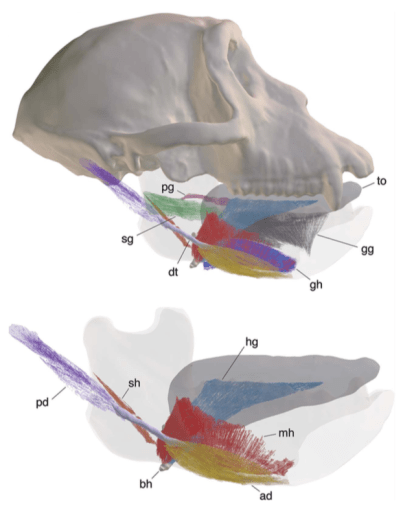
Advances in CT Technologies for LifeScience and Natural History Museum Collections
The Museum für Naturkunde and YXLON are partnering to deliver an Imaging Technology Symposium, June 12–13th 2019 in Berlin!
The symposium will bring together users of CT technology from biology, morphology, paleontology, zoology, and related areas to share their expertise and knowledge about exciting developments in biological imaging. A hands-on session by YXLON with their FF35 CT scanner (on June 14th) will follow the 2-day symposium.
The scientific program will consist of lectures from international scientists in the field as well as practically focused, hands-on and workshop sessions.
Interested? See more and register at: https://www.yxlon.com/news-events/events/events/2019-imaging-technology-symposium