
Avian vascular injection images by diceCT co-author Scott Echols and colleague Scott Birch were recently recognized as winners of the 2017 Wellcome Image Awards.
Congrats to both Scotts and kudos on the fabulous images—keep up the great work!

Avian vascular injection images by diceCT co-author Scott Echols and colleague Scott Birch were recently recognized as winners of the 2017 Wellcome Image Awards.
Congrats to both Scotts and kudos on the fabulous images—keep up the great work!

“Here we present a method that generates high quality iodine contrasted CT images from embryonic soft tissue. This method has two primary advantages over previous methodologies. First, it allows extraction of high quality nucleic acids following the CT scan, generating a 1:1 correspondence between scan and gene expression or DNA profile. Second, it minimizes the shrinkage artifacts associated with fixation and contrasting. The ability to relate a single sample to its genetic profile will allow study of both partially penetrant models and also increase our understanding of how genotype and phenotype are directly related.”
– Lead author, Rebecca Green (Hallgrímsson Lab)
Go read more at Developmental Dynamics!
“A three-dimensional model of the wing musculature of the sparrowhawk (Accipiter nisus) was built using μCT and a 3% iodine-buffered formalin solution to test the ability of the technique for visualising wing musculature and obtaining quantitative data of muscle geometry. This model allows the identification of the individual muscles comprising the avian wing and can be useful for further biomechanical analysis of flight.”
– Lead Author, Fernanda Bribiesca-Contreras
Go read more about the project at PeerJ!

“Tour Through the Brain of Python,” a diceCT visualized video fly-through of a Macklot’s python head by Paul Gignac and Nathan Kley, is a winner in the fifth annual FASEB BioArt Competition!
It’s so important to seize opportunities to share the wonder of discovery with the public. The BioArt images showcase the beauty of scientific research and are a great place to start the conversation —Hudson Freeze, PhD, FASEB President.
The Federation of American Societies for Experimental Biology holds an annual BioArt Competition to help engage Members of Congress and the general public about the immense value of biomedical and bioimaging research in the United States and the need for sustained support of federal funding agencies that facilitate life science and biomedical studies. To celebrate the competition, winning entries will be exhibited at the National Institutes of Health and online at FASEB.org.
“We used high precision computed tomography (CT) and traditional radiography to study the nasal conchae, complex structures within the nasal cavity that condition air via countercurrent heat exchange. Concha size differed between 2 subspecies of Song Sparrow (Melospiza melodia) that inhabit climatically distinct habitats, suggesting adaptation to local climates. The conchae and external bill are nested structures that were positively related in size and play functionally related roles in thermoregulation, therefore suggesting phenotypic integration. We hypothesize that the typically deeper and wider bill of the dune subspecies has evolved, at least in part, to accommodate larger conchae.”
– Lead author, Raymond Danner
Head over to The Auk to see the the published image sets!

 For those diceCT users working STABILITY protocols into your specimen preparation regimes, we’ve been hearing reports that your thermally-triggered initiator, VA-044 (2,2′-Azobis[2-(2-imidazolin-2-yl)propane]dihydrochloride), may be difficult to source. It tends to be sold in larger volumes than researchers practically require for STABILITY, which has made it difficult to purchase while leaving much of the chemical unused on researcher’s shelves.
For those diceCT users working STABILITY protocols into your specimen preparation regimes, we’ve been hearing reports that your thermally-triggered initiator, VA-044 (2,2′-Azobis[2-(2-imidazolin-2-yl)propane]dihydrochloride), may be difficult to source. It tends to be sold in larger volumes than researchers practically require for STABILITY, which has made it difficult to purchase while leaving much of the chemical unused on researcher’s shelves.
Dr. Vera Weisbecker (University of Queensland, Queensland, Australia) has recently offered to share some of her supply with STABILITY users, who might not be able to get a hold of it from Wako Specialty Chemicals or elsewhere. Please email her directly if you are interested.
We, @DiceCT, would like to offer an enormous thank you to Dr. Weisbecker for her generosity and openness, in particular, as well as for advancing STABILITY for our larger community!
A diceCT visualized ratsnake head and brain is a finalist in the 2016 Royal Society of London Scientific Photography Competition! This piece and many other contributions of remarkable scientific artwork are now part of a display at the Royal Society of London Online.
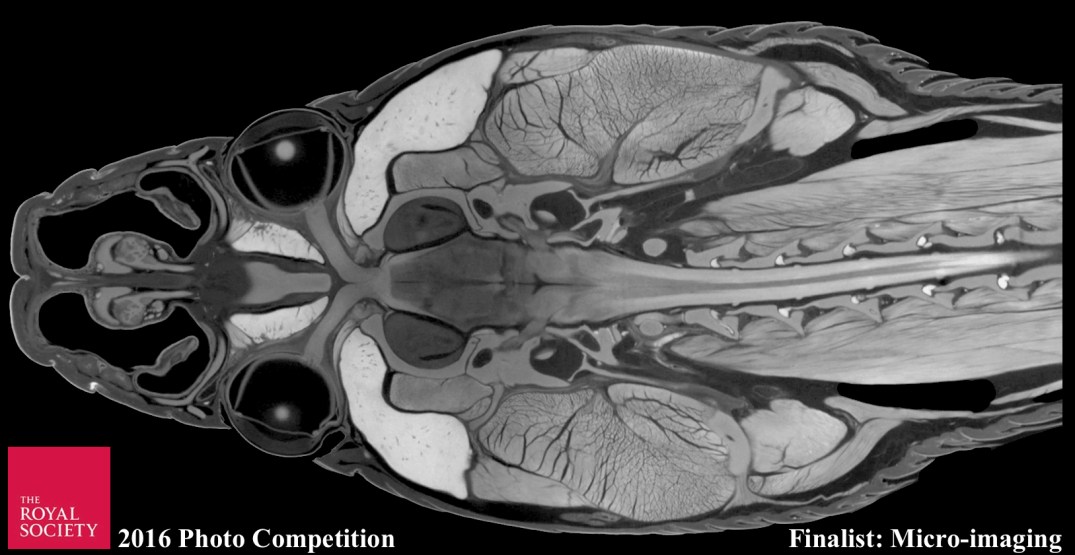
The competition celebrates the power of photography to communicate science and the role images play in making science more accessible to a wide audience. To view featured photos at the Open House exhibition (9/17–9/18/2016), visit The Royal Society of London.
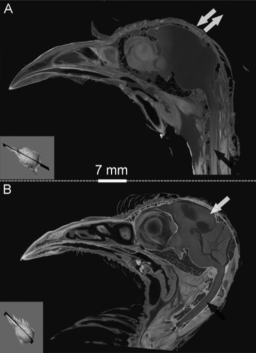
“I2KI-formalin (iodine-potassium iodine-formalin) treatment can obscure the natural X-ray contrast of the bone due to neighboring soft-tissues during staining of avian cephalic material. By contrast, we found the I2E (iodine-ethanol) treatment can increase the X-ray opacity of not only soft tissues (e.g., muscles), but also the bone. Mathematical simulations suggest that remarkably different results from I2E and I2KI-formalin staining are due to different partition coefficients and retardation factors of tissues, fixation effects, and distinct iodine diffusion and sorption patterns. We also found a clear positive relationship between glycogen concentration and grayscale values measured within muscle, epithelia, nervous tissues, and glands regardless of the staining solution used.”
–Lead Author, Zhiheng Li
Head over to the Journal of Experimental Zoology to download the pub!
By Aki Watanabe (@akiopteryx)
Contrast limited adaptive histogram equalization (CLAHE) is a procedure for enhancing local contrast in an image or stack of images. In contrast to standard histogram equalization that applies single formula for enhancing contrast across the entire image, CLAHE applies multiple equalizations within partitions of an image, resulting in more localized and subtle contrast enhancements. This results in digital contrast enhancement that is not dominated by overly deep blacks or excessively bright whites.
For diceCT, CLAHE is very useful for improving edge recognition for digitally segmenting regions of interest (ROI) based on your CT data. It is particularly helpful when applied to sub-optimally stained specimens.
CLAHE is implemented in FIJI (ImageJ) and the script is available freely and openly. To perform it on a stack of CT images:
Here is a before and after example to illustrate how CLAHE enhances diceCT images.

CLAHE processes image stacks fairly quickly, so I recommend trying it with all diceCT image stacks. Generally, it improves edge recognition for all 3-D rendering programs, thus, greatly reducing the time spent on segmenting ROIs. As mentioned above, CLAHE is particularly useful for sub-optimally stained specimens, which is helpful when one cannot devote time or reserve frequent CT scanning sessions for checking and optimizing the stain concentration and duration.
Potential issues with CLAHE include the increase in file size associated with CT data from having both original and modified image stacks. In addition, CLAHE may accentuate unwanted artifacts like beam hardening, so there is further motivation to minimize such scanning artifacts.
Those interested in learning more about the method can visit Wikipedia and read through the original article (Zuiderveld, 1994).
Modified from Wong, Spring, & Henkelman (2013)
**UPDATE: Also see the June 26, 2017 edition of Scientific Reports for the full study
Mix all ingredients together. Ingredients should be kept on ice during preparation of the solution to prevent premature polymerisation.
Immerse specimens in hydrogel solution for 1-2 weeks at 4° C. Larger specimens should be immersed for longer, and specimen type may be important too (e.g., lizards probably take longer than tadpoles). Users should be aware that the immersion time here is longer than for the original STABILITY protocol (Wong et al., 2013). This may be due to longer fixation in formalin, which seems to use up some biomolecule binding sites.
Any type of vegetable/nut/canola oil can be used for curing (approximately 3 ml). Carefully pour the oil on the surface of the hydrogel solution to form an airtight layer. Some researchers use a vacuum pump nitrogen replacement step, but we found the oil easier. The vials are placed in a water bath set at 37º C for three hours, triggering polymerisation of the gel. After polymerisation, remove excess gel (now solidified) with clean gloves and lint-free wipes, revealing the specimen as a gel/tissue hybrid. Specimens were then placed directly into iodine solution.

For staining we used 1.75% IKI solution, for about 30% longer than we would normally stain—but this depends on specimens and needs to be refined a bit more. Lower-concentration solutions may yield even less shrinkage. Aki Watanabe (pers. comm.) reported slower uptake of iodine at room temperature, which he found advantageous for a more controlled stain. We also found that staining in the fridge reduced the number of high-density “pockets” of iodine producing interference during scanning.

Please feel free to use our protocol. If you do so, we would appreciate a citation:
Carlisle, A., Weisbecker, V. (2016) A modified STABILITY protocol for accurate retrieval of soft-tissue data from micro-CT scans of IKI-stained specimens. Published online at https://dicect.com/2016/08/09/stability/, August 09, 2016.
Reference:
Wong, M. D., Spring, S., & Henkelman, R. M. (2013). Structural stabilization of tissue for embryo phenotyping using micro-CT with iodine staining. PloS ONE, 8(12), e84321. [doi:10.1371/journal.pone.0084321]
Method presented at the 11th Meeting of the International Congress on Vertebrate Morphology